|
4/19/2023 0 Comments Ich guidance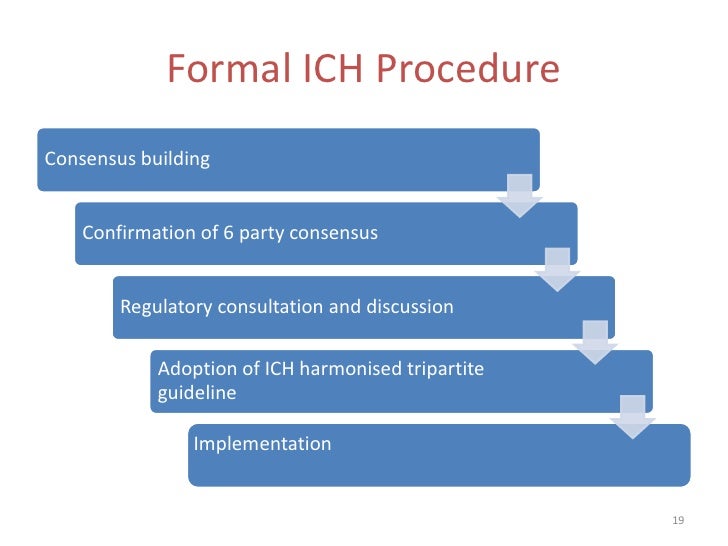 
The Extractables and Leachables Safety Information Exchange (ELSIE) Consortium, made up of 33 biopharma/medical device companies, is also active in this area and aims to provide expert input to efforts such as USP’s new chapter and the in-progress ICH Q3E, through working groups and formal commenting processes as well as white papers, workshops, and other public meetings. Industry experts say that questions about how this upcoming ICH guidance will align with the USP and BioPhorum documents are yet to be answered. A final concept paper for ICH Q3E was published in 2020 (4). The International Council for Harmonisation (ICH) is working on a document related to E&L testing. 
The analysis method depends on this risk level. USP’s accompanying procedural chapter became official on (3).Ī key aspect of the BioPhorum and new USP documents is the use of risk assessment for end-users to determine the risk level of a component in a particular process. USP General Chapter Plastic Components and Systems Used to Manufacture Pharmaceutical Drug Products and Biopharmaceutical Drug Substances and Products was published in 2022 (2), but the date for it to come into effect was postponed to 2026. The purpose of the extractables testing information package, says BioPhorum, is “to allow the SUS end-user to rigorously estimate the types and amounts of leachables that could be generated by the SUS component during its intended use” (1).Īnother set of standard methods was recently published in the United States Pharmacopeia (USP). SUS suppliers can use the protocol to measure the extractables in representative components. Because the organization was previously known as BioPhorum Operations Group (BPOG), the protocol is often called the BPOG protocol. Standard methods facilitate comparison between suppliers.īioPhorum, for example, first published a standardized extractables protocol in 2014, which was updated in 2020 (1). Industry organizations and consortiums have worked to standardize analysis protocols that can be used throughout the supply chain. As polymer single-use systems (SUS) are increasingly being used in biopharmaceutical manufacturing, there is a growing need to analyze E&L of this equipment. Processing equipment is not exempt, however. Container-closure systems, which contact the drug product during long-term storage, are often higher risk than process equipment, which has a short product-contact time. Dorival-García et al., Talanta 219, 121198 (2020).Įxtractables and leachables (E&L) must be analyzed for any product-contact material to determine the risk of leachable compounds contaminating a drug product. 
The use of ASE for material selection follows industry best practices for conducting controlled extraction studies. NIBRT researchers have also implemented a method based on accelerated solvent extraction (ASE) and HRMS for plastic raw material extractables characterization, which is a crucial first step in assessing suitability for an intended use (3). The researchers noted that the types of additives or manufacturing processes for the plastic bags could change, which could change the nature of E&L. NIBRT researchers found that polymer type and manufacturing date were the largest contributors to the different E&L profiles found in the different bags. Bags were extracted according to the BioPhorum Operations Group guidelines (2), and the extracts were analyzed using high-resolution mass spectrometry (HRMS) methods. The National Institute for Bioprocessing Research and Training (NIBRT) assessed the extractables and leachables (E&L) in 34 single-use bags used for cell cultivation in biopharmaceutical manufacturing the bags were from different manufacturers and represented eight different types of multilayer films (1).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
